Metals are exceptional conductors of heat and electricity due to their unique atomic structure and bonding characteristics. This property makes metals integral to various industrial and technological applications. Understanding the reasons behind the high conductivity of metals provides valuable insights into their role in modern society.

Credit: www.snexplores.org
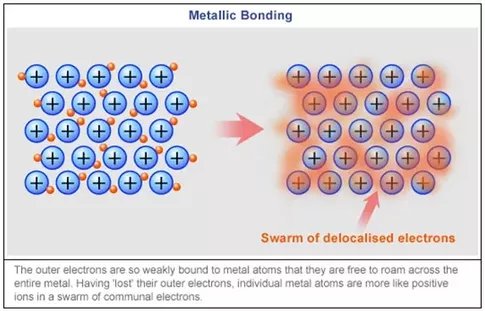
Atomic Structure of Metals
The atomic structure of metals contributes significantly to their excellent conductivity. Metals are composed of closely packed atoms arranged in a lattice structure. The outermost electrons of metal atoms are not strongly bound to any particular nucleus, allowing them to move freely throughout the metal. This free movement of electrons enables metals to conduct heat and electricity with minimal resistance.
Electron Mobility in Metals
Electron mobility is a key factor in determining the conductivity of metals. When energy, in the form of heat or electrical potential, is applied to a metal, the free electrons within the metal lattice gain kinetic energy and move rapidly. This movement of electrons facilitates the transfer of heat and the flow of electrical current. The availability of numerous free electrons in the metallic structure allows for efficient conduction of both heat and electricity.
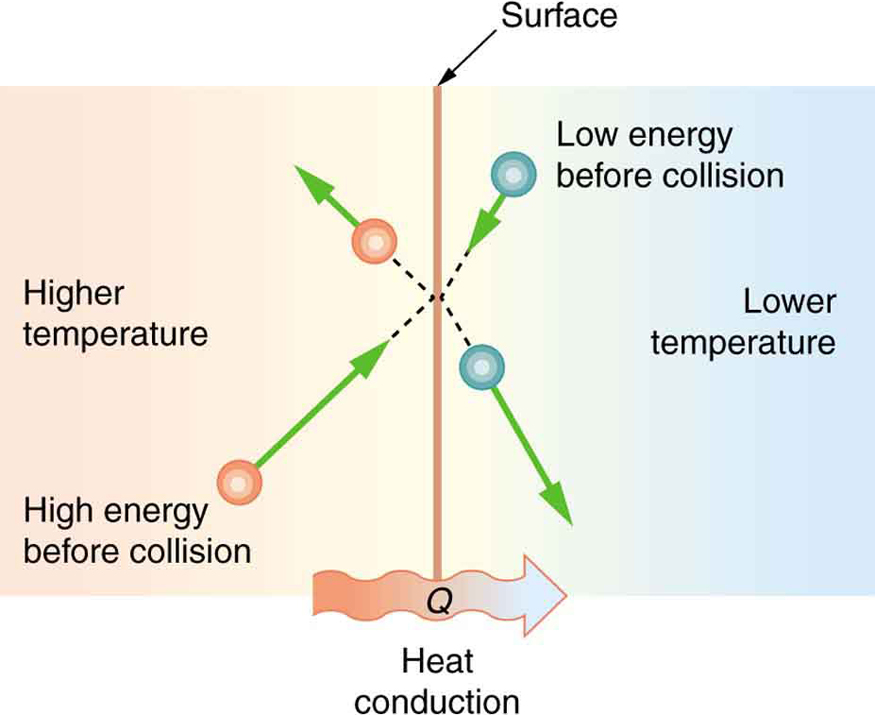
Thermal Conductivity of Metals
Metals possess high thermal conductivity, meaning they can efficiently transfer heat. As heat is applied to a metal, the free electrons absorb this energy and transfer it throughout the material. The rapid movement of electrons enables the heat to be conducted quickly, making metals effective conductors of thermal energy. This property is utilized in various applications, such as in the construction of cooking utensils, heat exchangers, and electrical wiring.
Electrical Conductivity of Metals
Similarly, the high electrical conductivity of metals results from the mobility of free electrons. When an electric potential is applied to a metal, the free electrons move in response to this potential difference, creating an electric current. This seamless movement of electrons allows for efficient electrical conduction, making metals ideal for use in electrical wiring, power transmission, and electronic components.
Comparison with Non-Metals
Unlike metals, non-metals lack the abundance of free electrons necessary for efficient heat and electrical conduction. The tightly bound electrons in non-metals restrict their mobility, leading to poor thermal and electrical conductivity. This stark contrast highlights the significance of the unique electron arrangement in metals, which enables them to excel as conductors of heat and electricity.
Factors Influencing Conductivity
Several factors can influence the conductivity of metals, including temperature and impurities. With an increase in temperature, the movement of atoms and lattice vibrations can impede the free electron mobility, leading to a decrease in conductivity. Additionally, impurities and alloying elements can affect the electron mobility within the metal lattice, thereby influencing its overall conductivity.
Applications of Metal Conductivity
The exceptional conductivity of metals is harnessed in a wide range of practical applications. From electrical wiring and power generation to industrial heating and cooling systems, metals play a pivotal role in enabling the efficient transfer of heat and electricity. Their conductivity also underpins the functionality of electronic devices, telecommunications infrastructure, and various manufacturing processes.
Frequently Asked Questions On Why Are Metals Good Conductors Of Heat And Electricity : Revealing The Secrets
Faq 1: Why Are Metals Good Conductors Of Heat And Electricity?
Metals are good conductors of heat and electricity due to their delocalized electrons, which can easily move and transfer energy.
Faq 2: How Do Metals Conduct Heat And Electricity?
Metals conduct heat and electricity through the movement of their free electrons, which flow and transfer energy from high to low temperatures or from areas of high to low electrical potential.
Faq 3: What Makes Metals Better Conductors Than Non-metals?
Metals are better conductors than non-metals because they have more free electrons that are mobile and can carry heat and electrical energy efficiently, while non-metals lack these free electrons.
Faq 4: Are All Metals Equally Good Conductors?
No, not all metals are equally good conductors. Some metals, such as copper and silver, have a higher conductivity due to their abundance of free electrons, while others like iron and aluminum have slightly lower conductivity but are still considered good conductors.
Conclusion
In conclusion, the exceptional conductivity of metals is attributed to their unique atomic structure, abundant free electrons, and the resulting efficient electron mobility. This inherent property enables metals to effectively conduct heat and electricity, making them indispensable in our daily lives and various industries. Understanding the underlying factors contributing to the conductivity of metals provides valuable insights that continue to drive technological advancements and innovation.